你鸭的是买药的吗?Brazil scientists say Sinovac vaccine CoronaVac’s has proven effective in late-stage trials: Report
Brazil is the first country to complete late-stage trials of CoronaVac, which is also being tested in Indonesia and Turkey.
您正在使用一款已经过时的浏览器!部分功能不能正常使用。
请尝试升级或使用 其他浏览器。
请尝试升级或使用 其他浏览器。
中国第二款,即科兴疫苗,土耳其第三期83.5%。10 weeks 内可生产出针对变异病毒的产品。2021.6.1 WHO批准
- 主题发起人 向问天
- 开始时间
billwanhua
本站元老
- 注册
- 2005-07-07
- 消息
- 17,284
- 荣誉分数
- 5,728
- 声望点数
- 373

Covid cases rise across more than half of the U.S. as country races to vaccinate
White House Chief Medical Advisor Dr. Anthony Fauci urged states last week not to declare victory prematurely.
对,效果肯定是有的。只是,数据也都摆在那儿。智利、土耳其,等等几十个国家,都还在加大力度购买中国疫苗。如果没有效果,那是他们太蠢了。如果他们不蠢,就是你们太蠢。
如果智利人打了中国疫苗后,这些人的感染率还是一样,肯定就不会再加大采购。比如南非,发现阿斯利康不行,立即退货,是不可能加大采购的。
Chile receives 2 million doses of COVID-19 vaccine
A new shipment carrying 2 million doses of the Sinovac COVID-19 vaccine arrived in Chile on Sunday
Written by: Associated Press Television News
Updated Mar 22, 2021 7:45 AM
补充一下,土耳其目前疫苗接种率是15.5%
最后编辑:
billwanhua
本站元老
- 注册
- 2005-07-07
- 消息
- 17,284
- 荣誉分数
- 5,728
- 声望点数
- 373
Hold your horse. 美国打那么多95%有效率的疫苗,英国的AZ疫苗都多得没有用处,感染率不降反升,你能得到什么结论?按照你们逻辑是不是加拿大打的AZ疫苗效率更高?
There are too many factors to weight in
There are too many factors to weight in
- 注册
- 2005-11-23
- 消息
- 30,005
- 荣誉分数
- 7,409
- 声望点数
- 373
Hold your horse. 美国打那么多95%有效率的疫苗,英国的AZ疫苗都多得没有用处,感染率不降反升,你能得到什么结论?按照你们逻辑是不是加拿大打的AZ疫苗效率更高?
There are too many factors to weight in
英国阳性率下降最为惊人,从1月初的12.4到现在的0.4%,印度最近几乎失控。美国的福奇已经的不停发警告了。
billwanhua
本站元老
- 注册
- 2005-07-07
- 消息
- 17,284
- 荣誉分数
- 5,728
- 声望点数
- 373
美国接种率多少?
- 注册
- 2005-11-23
- 消息
- 30,005
- 荣誉分数
- 7,409
- 声望点数
- 373
一针41.2%,2针3.4%美国接种率多少?
billwanhua
本站元老
- 注册
- 2005-07-07
- 消息
- 17,284
- 荣誉分数
- 5,728
- 声望点数
- 373
2针34% ?一针41.2%,2针3.4%
- 注册
- 2005-11-23
- 消息
- 30,005
- 荣誉分数
- 7,409
- 声望点数
- 373
billwanhua
本站元老
- 注册
- 2005-07-07
- 消息
- 17,284
- 荣誉分数
- 5,728
- 声望点数
- 373
3.4%
1+2针总数44.6%
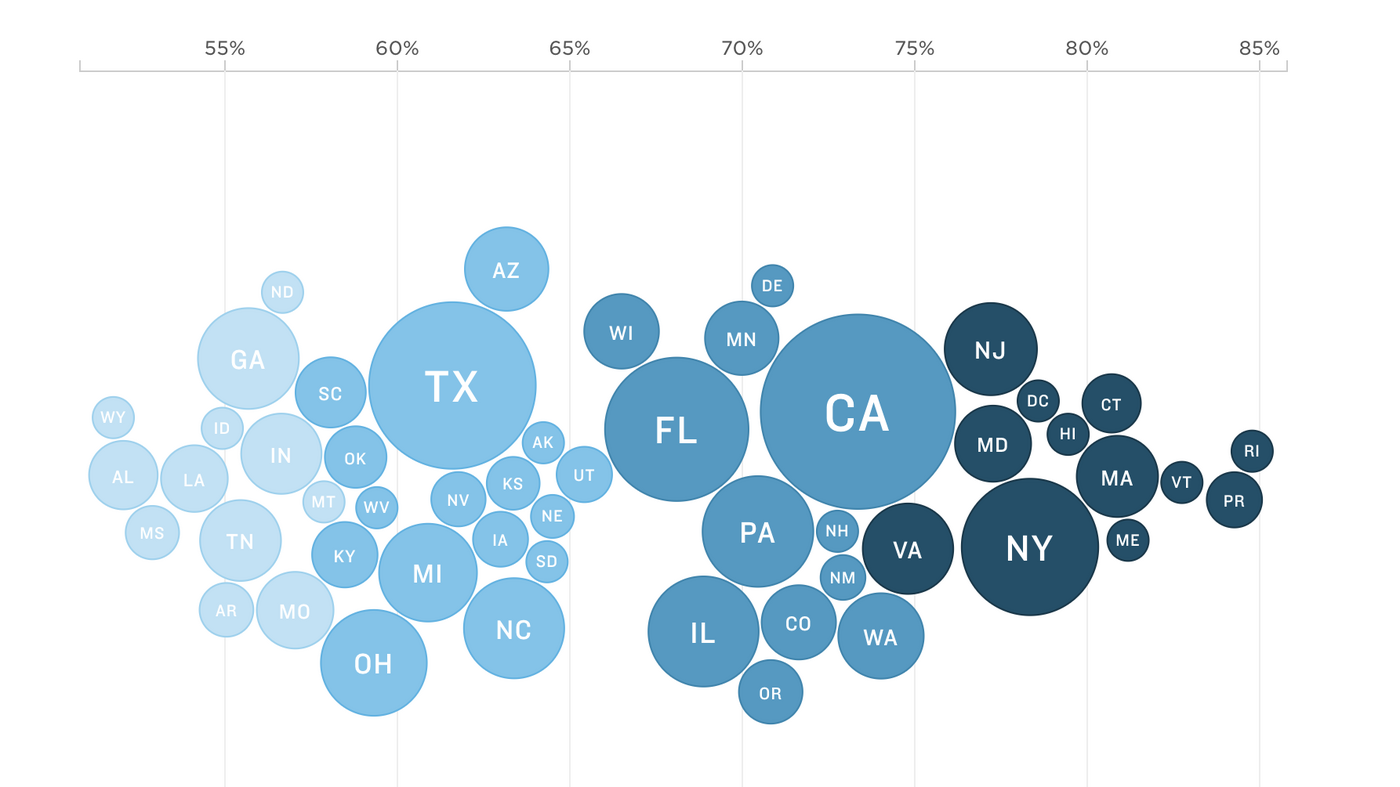
How are the COVID-19 vaccine and booster campaigns going in your state?
The U.S. is striving to vaccinate as many people as possible against COVID-19 — and keep them up-to-date with boosters. But some states are lagging behind. See how yours is faring.
你的数据不对,13.5%, 24.9%
- 注册
- 2005-11-23
- 消息
- 30,005
- 荣誉分数
- 7,409
- 声望点数
- 373
美国接种率多少?
Sorry 我看错了,以为你问英国。
How are the COVID-19 vaccine and booster campaigns going in your state?
The U.S. is striving to vaccinate as many people as possible against COVID-19 — and keep them up-to-date with boosters. But some states are lagging behind. See how yours is faring.www.npr.org
你的数据不对,13.5%, 24.9%
国家 Country | 百人中接种总针数 Percentage | 总接种针数 (百万) Total Number (millions) | 接种第一针人口百分数 Percentage | 接种第一针人数 (百万) Total Number (millions) | 已接种两针者百分数Fully Vaccinated % | 已接种两针人数(百万) Fully Vaccinated (millions) | 阳性率 Positive Rate % | 日期 Date | 阳性率 Positive Rate % | 变化率 Change Percentage |
以色列 Israel | 112.99 | 9.78 | 59.9 | 5.19 | 53.1 | 4.59 | 10.3 | Feb. 1 | 2.0 | - 81% |
阿联酋 UAE | 74.56 | 7.37 | 35.2 (Feb. 23) | 3.48 (Feb. 23) | 22.1 | 2.19 | 2.4 | Jan. 19 | 0.9 | - 63% |
加拿大 CANADA | 10.86 | 4.10 | 9.2 | 3.46 | 1.7 | 0.633 | 8.9 | Jan. 29 | 3.5 | - 61% |
英国 UK | 44.6 | 30.28 | 41.2 | 28.00 | 3.4 | 2.28 | 12.8 | Jan. 4 | 0.4 | - 97% |
美国 USA | 37.83 | 126.51 | 24.8 | 82.77 | 13.4 | 44.91 | 14.6 | Jan. 7 | 5.8 | - 60% |
中国 China | 5.21 | 74.96 | ? | |||||||
印度 India | 3.51 | 48.49 | 2.9 | 40.63 | 0.6 | 7.86 | 1.9 | Feb. 1 | 3.9 | + 105% |
世界 World | 5.88 | 458.13 | 3.5 | 271.12 | 1.3 | 104.12 |
|
- 注册
- 2005-11-23
- 消息
- 30,005
- 荣誉分数
- 7,409
- 声望点数
- 373
Sorry 我看错了,以为你问的是英国。
How are the COVID-19 vaccine and booster campaigns going in your state?
The U.S. is striving to vaccinate as many people as possible against COVID-19 — and keep them up-to-date with boosters. But some states are lagging behind. See how yours is faring.www.npr.org
你的数据不对,13.5%, 24.9%
国家 Country | 百人中接种总针数 Percentage | 总接种针数 (百万) Total Number (millions) | 接种第一针人口百分数 Percentage | 接种第一针人数 (百万) Total Number (millions) | 已接种两针者百分数Fully Vaccinated % | 已接种两针人数(百万) Fully Vaccinated (millions) | 阳性率 Positive Rate % | 日期 Date | 阳性率 Positive Rate % | 变化率 Change Percentage |
以色列 Israel | 112.99 | 9.78 | 59.9 | 5.19 | 53.1 | 4.59 | 10.3 | Feb. 1 | 2.0 | - 81% |
阿联酋 UAE | 74.56 | 7.37 | 35.2 (Feb. 23) | 3.48 (Feb. 23) | 22.1 | 2.19 | 2.4 | Jan. 19 | 0.9 | - 63% |
加拿大 CANADA | 10.86 | 4.10 | 9.2 | 3.46 | 1.7 | 0.633 | 8.9 | Jan. 29 | 3.5 | - 61% |
英国 UK | 44.6 | 30.28 | 41.2 | 28.00 | 3.4 | 2.28 | 12.8 | Jan. 4 | 0.4 | - 97% |
美国 USA | 37.83 | 126.51 | 24.8 | 82.77 | 13.4 | 44.91 | 14.6 | Jan. 7 | 5.8 | - 60% |
中国 China | 5.21 | 74.96 | ? | |||||||
印度 India | 3.51 | 48.49 | 2.9 | 40.63 | 0.6 | 7.86 | 1.9 | Feb. 1 | 3.9 | + 105% |
世界 World | 5.88 | 458.13 | 3.5 | 271.12 | 1.3 | 104.12 |
- 注册
- 2012-09-04
- 消息
- 64,350
- 荣誉分数
- 11,677
- 声望点数
- 1,373
Sinopharm, Sinovac COVID-19 Vaccine Data Show Efficacy: WHO
By Reuters, Wire Service Content March 31, 2021GENEVA (REUTERS) - Chinese vaccine makers Sinopharm and Sinovac have presented data on their COVID-19 vaccines indicating levels of efficacy that would be compatible with those required by the World Health Organization, the chair of a WHO advisory panel said on Wednesday.
The WHO's Strategic Advisory Group of Experts (SAGE) hopes to issue recommendations on those vaccines by the end of April, its chair Alejandro Cravioto, told a Geneva news briefing.
"The information that the companies shared publicly at the (SAGE) meeting last week clearly indicates that they have levels of efficacy that would be compatible with the requirements that WHO has asked for this vaccine," Cravioto said, referring to the group of independent experts' closed-door meeting.
"That means about 50% (efficacy) and preferably close to or above 70% and of course, they have all the safety data to show that this vaccine would cause no harm in humans when used."
The vaccines would first require emergency use listing from the WHO or from what the agency considers to be a stringent regulatory authority before SAGE experts can make recommendations about their use, he added.
WHO spokeswoman Margaret Harris said earlier this month that Sinopharm and Sinovac vaccines could receive WHO emergency listing "quite soon".
No detailed efficacy data of Sinopharm's COVID-19 vaccine has been publicly released but its developer, Beijing Biological Products Institute, a unit of Sinopharm subsidiary China National Biotec Group (CNBG), said the vaccine was 79.34% effective in preventing people from developing the disease based on interim data. It has been approved in several countries including China, Pakistan and the UAE.
Sinovac's vaccine showed varied efficacy readings of between 50.65% and 83.5% based on trials from Brazil, Turkey and Indonesia.
The two China-made vaccines have seen strong demand from many developing countries which have limited access to shots made by rival Western drugmakers.
(Reporting by Stephanie Nebehay and Kate Kelland)
Copyright 2021 Thomson Reuters.
happy 2016
高级会员
- 注册
- 2016-01-12
- 消息
- 1,004
- 荣誉分数
- 354
- 声望点数
- 93
Sinopharm, Sinovac COVID-19 Vaccine Data Show Efficacy: WHO
By Reuters, Wire Service Content March 31, 2021
GENEVA (REUTERS) - Chinese vaccine makers Sinopharm and Sinovac have presented data on their COVID-19 vaccines indicating levels of efficacy that would be compatible with those required by the World Health Organization, the chair of a WHO advisory panel said on Wednesday.
The WHO's Strategic Advisory Group of Experts (SAGE) hopes to issue recommendations on those vaccines by the end of April, its chair Alejandro Cravioto, told a Geneva news briefing.
"The information that the companies shared publicly at the (SAGE) meeting last week clearly indicates that they have levels of efficacy that would be compatible with the requirements that WHO has asked for this vaccine," Cravioto said, referring to the group of independent experts' closed-door meeting.
"That means about 50% (efficacy) and preferably close to or above 70% and of course, they have all the safety data to show that this vaccine would cause no harm in humans when used."
The vaccines would first require emergency use listing from the WHO or from what the agency considers to be a stringent regulatory authority before SAGE experts can make recommendations about their use, he added.
WHO spokeswoman Margaret Harris said earlier this month that Sinopharm and Sinovac vaccines could receive WHO emergency listing "quite soon".
No detailed efficacy data of Sinopharm's COVID-19 vaccine has been publicly released but its developer, Beijing Biological Products Institute, a unit of Sinopharm subsidiary China National Biotec Group (CNBG), said the vaccine was 79.34% effective in preventing people from developing the disease based on interim data. It has been approved in several countries including China, Pakistan and the UAE.
Sinovac's vaccine showed varied efficacy readings of between 50.65% and 83.5% based on trials from Brazil, Turkey and Indonesia.
The two China-made vaccines have seen strong demand from many developing countries which have limited access to shots made by rival Western drugmakers.
(Reporting by Stephanie Nebehay and Kate Kelland)
Copyright 2021 Thomson Reuters.
(法新社)消息.
巴基斯坦总统阿尔维本月15日接种中国国药疫苗Sinopharm.在週一(29日),阿尔维在推特中公布确诊消息,指出他在接种首剂中国新冠疫苗后仍验出病毒阳性反应。除了总统阿尔维以外,巴基斯坦总理伊姆兰汗(Imran Khan)本月在注射过第1剂疫苗后,在3月20日也验出病毒阳性反应,目前正在私人住宅中隔离.伊姆兰汗的妻子以及执政党「巴基斯坦正义运动党」的两名高层也确诊阳性。
在週一(29日),阿尔维在推特中公布确诊消息,指出他在接种首剂中国新冠疫苗后仍验出病毒阳性反应。除了总统阿尔维以外,巴基斯坦总理伊姆兰汗(Imran Khan)本月在注射过第1剂疫苗后,在3月20日也验出病毒阳性反应,目前正在私人住宅中隔离.伊姆兰汗的妻子以及执政党「巴基斯坦正义运动党」的两名高层也确诊阳性。
- 注册
- 2012-09-04
- 消息
- 64,350
- 荣誉分数
- 11,677
- 声望点数
- 1,373
(法新社)消息.
巴基斯坦总统阿尔维本月15日接种中国国药疫苗Sinopharm.在週一(29日),阿尔维在推特中公布确诊消息,指出他在接种首剂中国新冠疫苗后仍验出病毒阳性反应。除了总统阿尔维以外,巴基斯坦总理伊姆兰汗(Imran Khan)本月在注射过第1剂疫苗后,在3月20日也验出病毒阳性反应,目前正在私人住宅中隔离.伊姆兰汗的妻子以及执政党「巴基斯坦正义运动党」的两名高层也确诊阳性。
在週一(29日),阿尔维在推特中公布确诊消息,指出他在接种首剂中国新冠疫苗后仍验出病毒阳性反应。除了总统阿尔维以外,巴基斯坦总理伊姆兰汗(Imran Khan)本月在注射过第1剂疫苗后,在3月20日也验出病毒阳性反应,目前正在私人住宅中隔离.伊姆兰汗的妻子以及执政党「巴基斯坦正义运动党」的两名高层也确诊阳性。
中国疫苗防止感染的有效率不是100%。
