- 注册
- 2005-11-23
- 消息
- 30,005
- 荣誉分数
- 7,409
- 声望点数
- 373
据2月份的报道,以色列辉瑞疫苗60万人,打了两针以后两周测(此时抗体最高),608人感染,占0.1%,21人住院,其中7人重症,都有基础病,3人中度,11人轻症。(法新社)消息.
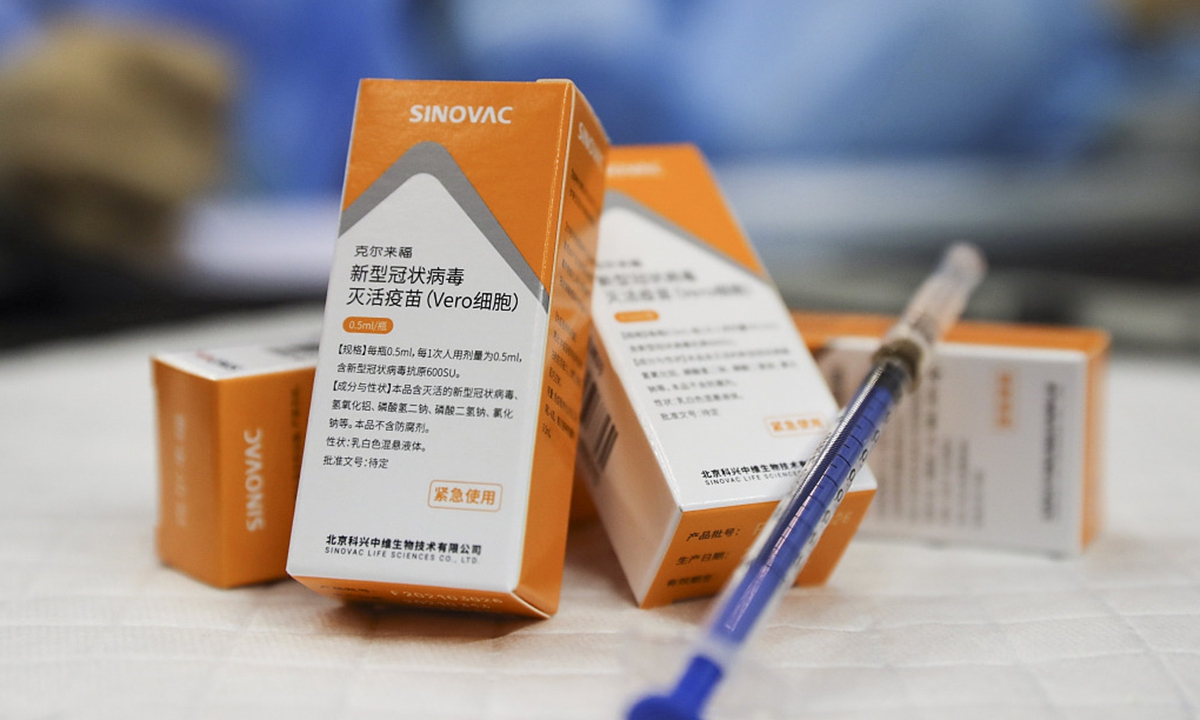
巴基斯坦总统阿尔维本月15日接种中国国药疫苗Sinopharm.在週一(29日),阿尔维在推特中公布确诊消息,指出他在接种首剂中国新冠疫苗后仍验出病毒阳性反应。除了总统阿尔维以外,巴基斯坦总理伊姆兰汗(Imran Khan)本月在注射过第1剂疫苗后,在3月20日也验出病毒阳性反应,目前正在私人住宅中隔离.伊姆兰汗的妻子以及执政党「巴基斯坦正义运动党」的两名高层也确诊阳性。
在週一(29日),阿尔维在推特中公布确诊消息,指出他在接种首剂中国新冠疫苗后仍验出病毒阳性反应。除了总统阿尔维以外,巴基斯坦总理伊姆兰汗(Imran Khan)本月在注射过第1剂疫苗后,在3月20日也验出病毒阳性反应,目前正在私人住宅中隔离.伊姆兰汗的妻子以及执政党「巴基斯坦正义运动党」的两名高层也确诊阳性。
4:08 p.m. ET, February 17, 2021
Pfizer vaccine is about 95% effective against Covid-19, Israeli data shows
From CNN's Amir Tal and Elizabeth Cohen
Data from more than 600,000 vaccinated Israelis shows the Pfizer vaccine is 95% effective, with only 21 vaccinated people ending up in the hospital with Covid-19, according to a press release from Maccabi Healthcare Services, Israel’s second largest healthcare provider.
The findings, while preliminary, suggest the vaccine remains remarkably effective in the real world and not just in the clinical trials conducted last year by Pfizer and BioNTech.
Maccabi looked at data from about 602,000 of its members who were at least one week past their second dose of the vaccine. Among them, 7 were hospitalized with severe symptoms, 3 with moderate symptoms, and 11 with mild symptoms.
All of the hospitalized members were over age 50, and half of them had a chronic health condition, such as morbid obesity, according to Ido Hadari, a Maccabi spokesman.
Among the 602,000 vaccinated members, tests showed that 608 became infected with Covid-19. Maccabi did not routinely test members, but of the 608, 35% chose to get tested because they had symptoms of Covid-19 and 65% chose to get tested because they had been exposed to someone with the virus.
Maccabi compared that infection rate with a group of 528,000 members who had not yet received the vaccine. In this control group, 20,621 people, or nearly 4%, were identified as having Covid-19.
Maccabi’s findings are similar to data released earlier this week by Israel’s largest healthcare provider, which found the vaccine was 94% effective. Pfizer’s clinical trial showed about the same efficacy.
Pfizer vaccine is about 95% effective against Covid-19, Israeli data shows